An Electronic Data Capturing (EDC) or Direct Data Capture (DDC) for collection of clinical data through computerized system helps accelerating and streamlining the data collection method by replacing the traditional paper based CRF, in an organized and standardized process. But mapping raw Electronic Data Capture (EDC) data to a Study Data Tabulation Model (SDTM) or Analysis Data Model (ADaM) standard is a very time consuming process.
As the standards are continuously evolving and new guidelines are coming up, the ability to collect and reuse the raw variables and mapping it with the standardized variables can help reduce time and efforts to standardize the study data.
The reduction in time and efforts for getting the data in the standardized format for all the studies will come from the ability to reuse the electronic data capturing templates which can be made as per the standard itself. Using the power of Machine Learning, algorithms can further help derive and predict possible creation of the templates.
Creating eCRF using the standard variables as per SDTM defined will take the data input in formatted manner. Using the algorithms to set the default values which are generally which can be common across the studies automates the system and reduces the user work.
Defining the variables in eCRF in such a way that can be reused and roles can be defined as per the review and authorization given to a particular designation. So, the data goes under the authorization cycle, which can do data cleaning and data review. The data can be divided as per role and designation of the person.
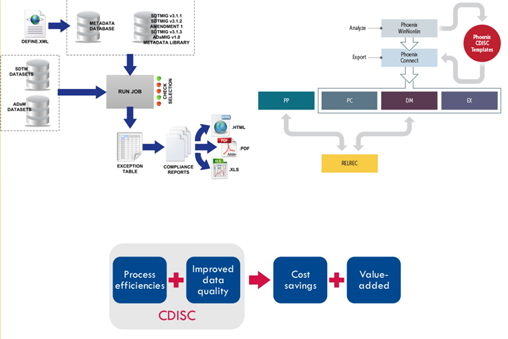
The data which is already captured in the standard SDTM format can be used further in Analysis Data and creating reviewer guides.
The capitalization of capturing the data in standard format for the CRO’s would be reduction in cost of buying a system which would be mapping the raw data to standardized data as the data captured itself would be in SDTM format.
We, have worked on such a system which generates the data in SDTM format. Which has a flexibility of having user defined naming conventions as per the sponsor’s requirement too.
System has easy flow and use of CDISC standards from CRF design to data collection, cleaning and finally exporting of the entire data to tabulation for analysis. Application is beneficial in terms of cutting cost and time followed up by improving data integrity with proper data mapping.
And having algorithms to set the predefined values to the variables which flows throughout the study which are reviewed and authorized.
BizNET is an end-to-end solution which caters to all the needs of clinical research project starting from Project Management, feasibility assessment, volunteer or patient enrollment using iris recognition technology, capture clinical trial data, multi-level review process, data compilation to data archiving.
Solution blends various integrated modules that prominently support clinical trial activities like EDC/DDC data collection, IWRS & drug randomization management, SDTM tabulation.
BizNET is a validated software, compliant with 21 CFR part 11 and GCP guidelines which provide a transparent and smooth process to regulatory bodies and sponsors.